About Us

Dongmai Medical Efficiency Engine
A trusted and integrated enabling partner for manufacturers
Reducing production costs: Leveraging Dongmai Medical's manufacturing matrix and integrated supply chain resources, we enable the sharing of production resources to effectively help medical device manufacturers reduce costs and improve efficiency.
Professional compliance assurance: Based on the intellectual property protection philosophy of not claiming ownership of clients' registration certificates, a mature Marketing Authorization Holder (MAH) quality management system, and CNAS laboratory internal quality control mechanisms, our CDMO model effectively reduces post-market compliance risks and management costs for medical device companies.
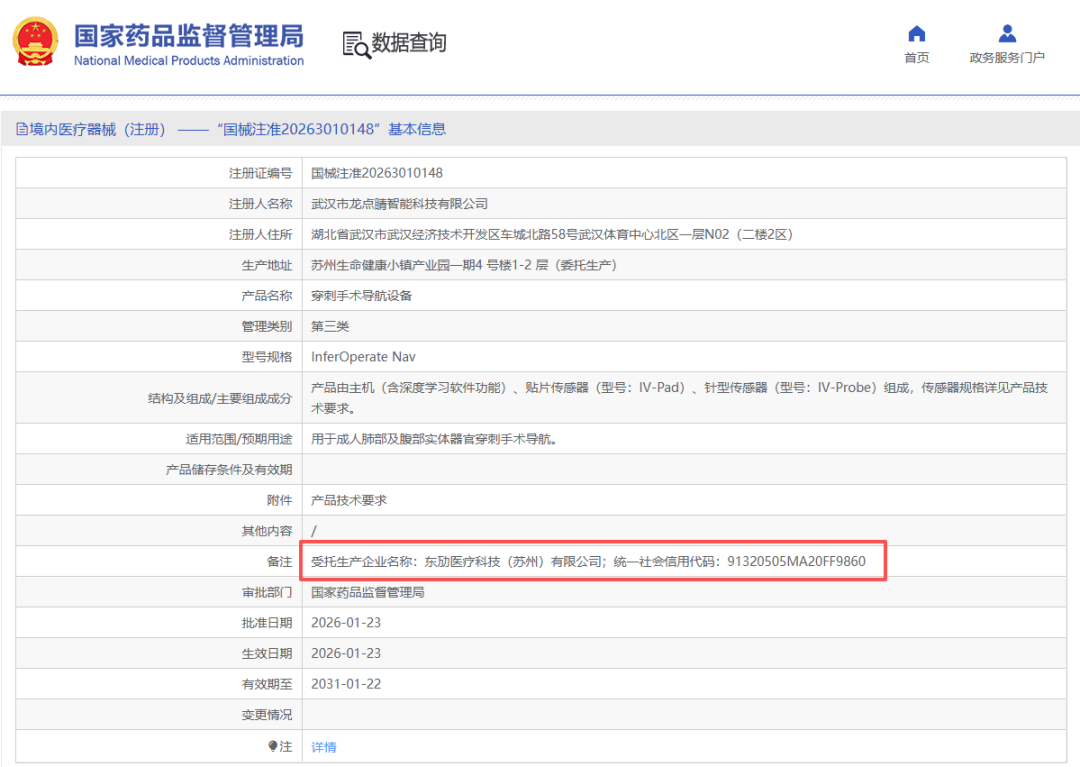
Improving registration efficiency: "Give us a proof-of-concept prototype, and we will deliver a registration certificate ready for mass production." By aligning product market positioning with regulatory requirements, Dongmai Medical accurately and efficiently designs medical device registration pathways. Focusing on compliance in both product deliverables and quality management systems, we facilitate engineering scale-up and manufacturing process transfer. Innovative measures, such as utilizing in-house self-testing reports for registration, effectively shorten the registration and approval cycle and accelerate time-to-market.
Source: Dongmai Medical Group CDMO+CRO